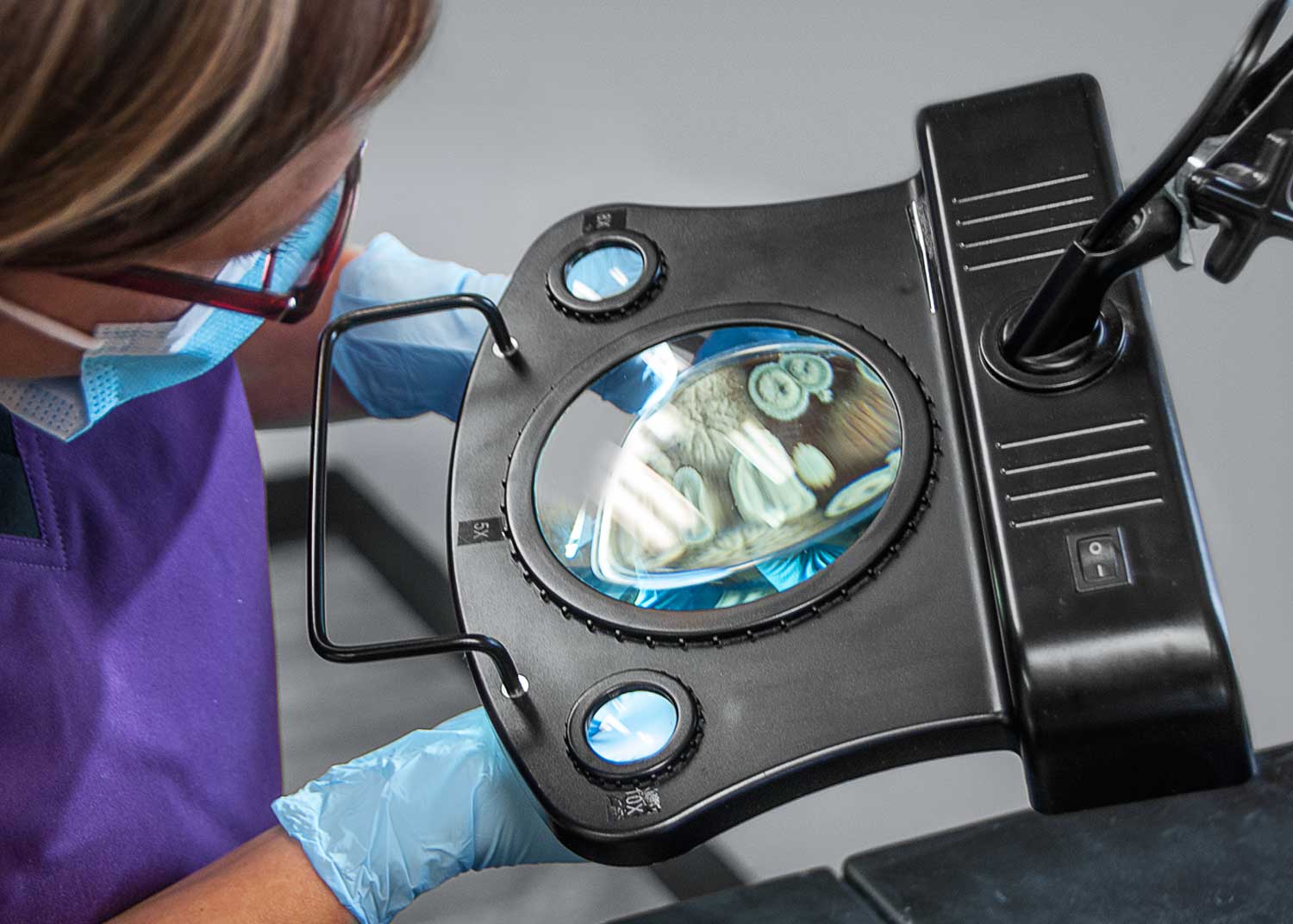
Laboratory Testing
Our ISO 13485:2016 Certified Laboratory Department is a well-equipped facility with a staff that holds a combined 90+ years of experience servicing the medical device industry. C.G. Laboratories, Inc. is committed to sound scientific principles. We focus on providing quality results, reliable turnaround times, responsive customer service while guiding the customer to understand their testing needs. Through ongoing competency training, as well as in-depth internal audits, we maintain the ability to deliver accurate results and quality customer service. We are prepared to meet all of your requirements – whether great or small and can serve as your full-time external laboratory or as a one-time service laboratory. We offer customized method development, along with assistance in designing special projects relating to non-routine analysis.
We understand our customers need prompt, accurate, affordable, and confidential testing. We also know that you prefer to work with scientists and technicians that are pleasant, courteous and dedicated to your needs. C.G. Laboratories, Inc. strives to accomplish these goals.
Our Laboratory Services Guide will give you a clear description of the testing that we currently provide. However, if you do not see the testing you require, give us a call. Our microbiologist will work with you to create the testing that is right for your medical devices.
C.G. Laboratories, Inc. should be your first choice for device testing, microbiological analysis, pharmaceutical analysis, regulatory testing, environmental monitoring and water testing.

Our Standards